January // February 2026 Progress Update
Biological scaling phenomena
In January of 2023, we outlined a plan to discover reprogramming payloads that could make old cells look and hopefully act young. We had yet to execute our first discovery screens, build our first AI systems, or even finish assembling our lab.
This January, we moved the first candidate medicine that emerged from that plan into large-scale manufacturing. We also discovered another payload that restores multiple youthful functions, built preclinical models in our third therapeutic program, and finalized an AI system that doubles our rate of discovery. These results have surpassed even our optimistic projections.
Recent highlights:
+1 payload that restores multiple youthful functions in old hepatocytes
0 → 20% manufacturing progress our first large-scale batch of drug candidate
0 → 4 candidate pharmacodynamic biomarkers
+50 payloads that make old hepatocytes look young
+19 payloads that rescue regulatory activity in T cells
+1 preclinical model of renal endothelial cell age
2X faster payload discovery with multi-task in silico reprogramming models
Restoring youthful metabolic function
Epigenetic marks on our DNA and surrounding proteins control which genes are turned on and off in each cell. Aging degrades these marks, and old cells lose the ability to perform obligate functions as a result. As one example, hepatocytes (Greek/biologist for “liver cell”) are less regenerative in the face of injury, less resilient to external damage, and less able to process metabolites.
NewLimit is discovering reprogramming payloads that rewrite these marks back to their youthful state. Each of our payloads activates a set of transcription factor (TF) genes that remodel the epigenome. In principle, payloads that effectively reprogram cell age could restore multiple youthful functions in old cells.
In February, we discovered another payload that displays this form of pleiotropy (Greek/biologist for “does multiple things”) in hepatocytes. After reprogramming with these TFs, old hepatocytes are both more resilient to damage from alcohol and more regenerative in the face of injury. The payload appears to have similar effects in both mouse and human hepatocytes, suggesting the biology is conserved across species.
When we began our discovery campaigns years ago, it was unclear if this form of multi-functional recovery was possible. We’ve now found multiple payloads that achieve this effect. Cell age can be reprogrammed while preserving cell type, and only a small set of transcription factors are required.
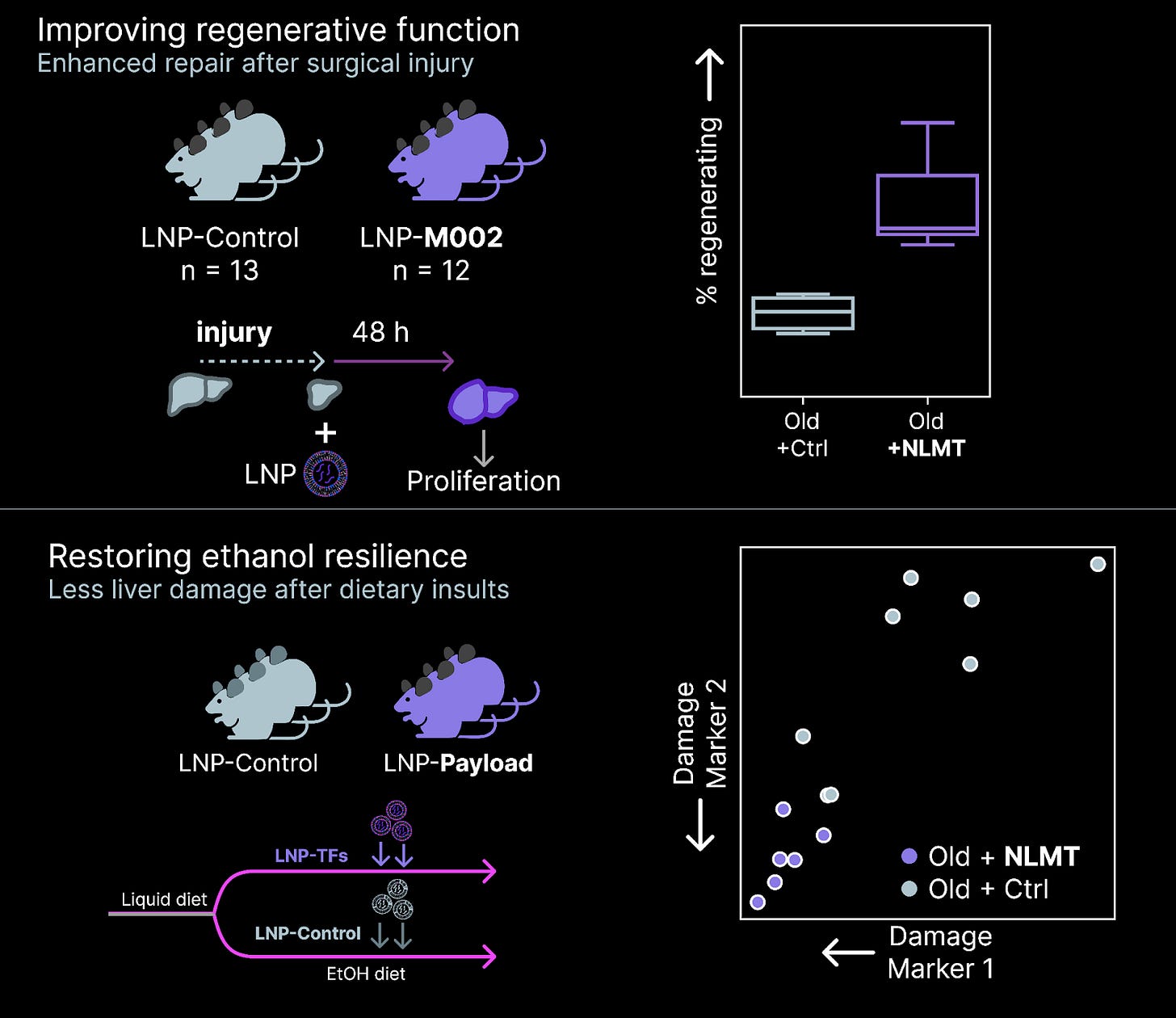
Inspired by results like these, we’ve been scaling our humanized liver reprogramming screens to discover new payloads. In the past months, we’ve landed 4 large-scale screens that revealed dozens of new payloads that restore youthful features in old human hepatocytes. These hits not only restored youthful gene expression, but also rescued regenerative function in this in vivo setting.
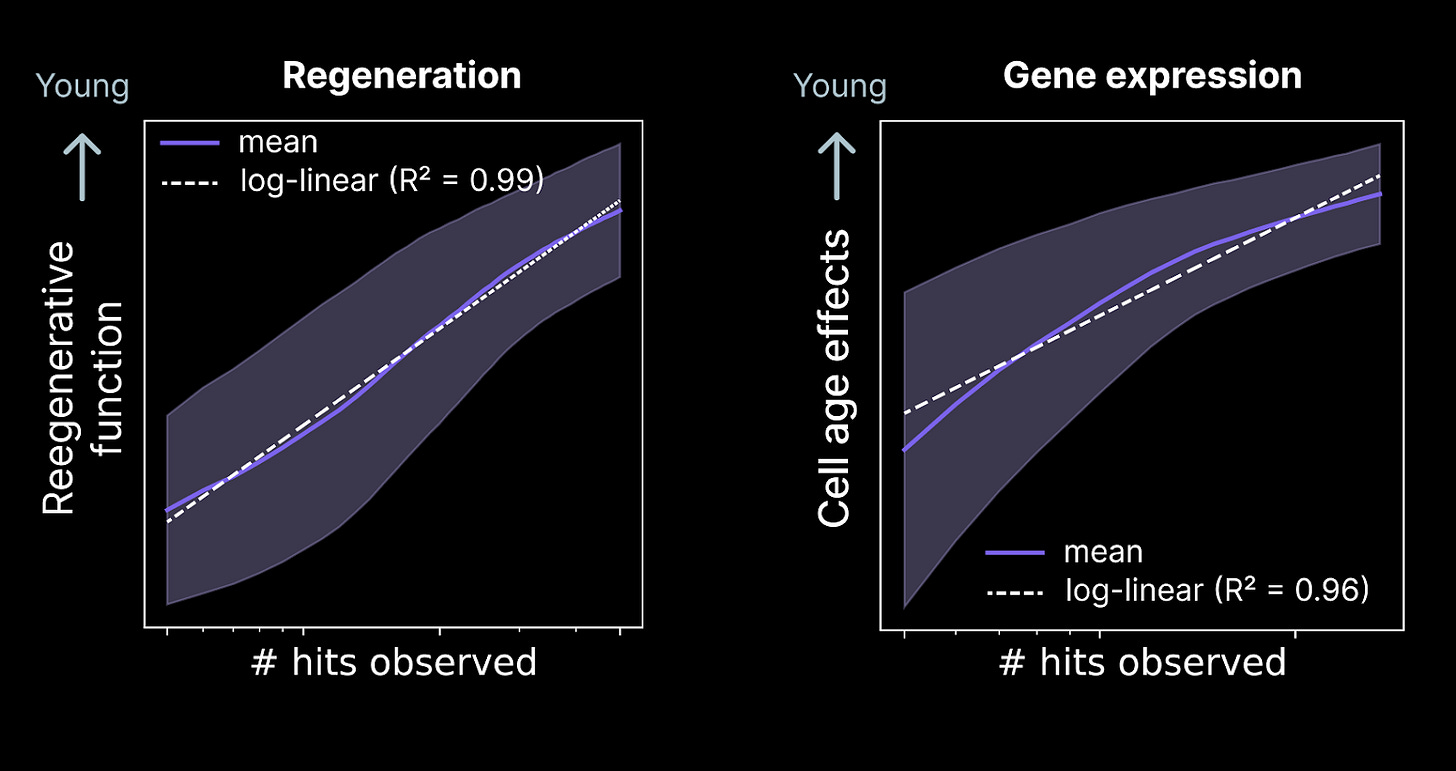
Therapeutics development typically involves discovering many promising candidates at the early stage, then carrying a small number of candidate medicines (e.g. 1-10) forward to clinical trials. In this setting, the quality of the top hits matters as much if not more than the number of hits we discover in total.
We’ve found that as we scale our discovery screens, the quality of the top k hits likewise continues to improve. This biological scaling law resembles the scaling properties of artificial intelligence systems and follows a log-linear trend. Due to this phenomenon, scaling our early stage discovery efforts is likely to result in better medicines when we enter development.
Artificial intelligence systems accelerate reprogramming discoveries
Discovering TF payloads that reprogram cell age is challenging because the number of possible payloads is uncountably large. There are about 1016 TF sets we might test, many orders of magnitude more than we can tractably test in the laboratory. In addition to building industrial scale experimental systems, we need to prioritize the experiments we run intelligently.
Humans struggle to reason through the complex prior information required for this prioritization task at scale. By contrast, modern artificial intelligence systems can achieve superhuman performance and help prioritize the most valuable experiments given only a sparse training set.
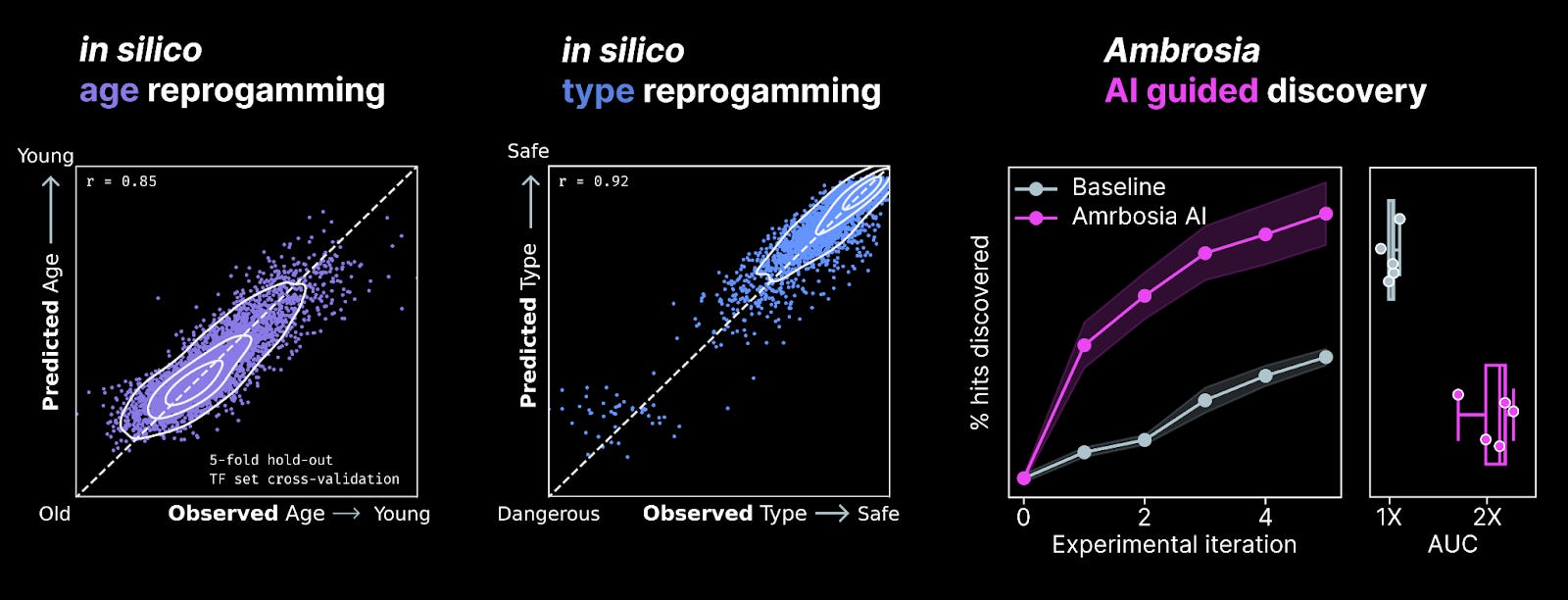
We’ve built a system we call Ambrosia to perform these reprogramming experiments in silico. In January, we upgraded our system to operate in a multi-task fashion, predicting several effects of reprogramming on cell state and function simultaneously. These in silico experiments now allow us to prioritize payloads based on multiple axes simultaneously. For instance, we prioritize payloads that are predicted to reverse cell age while also preserving cell type.
Our multi-task Ambrosia system is highly performant across a range of cell state and function prediction tasks. When we integrate the system with a lab-in-the-loop to perform discovery campaigns, we achieve our strongest ever performance and increase our rate of discovery >2X relative to the baseline. Every time we launch a screen at NewLimit – multiple times per month – we’re able to make more discoveries per dollar as a result.
We’re working now to expand the palette of functional effects our system can both predict and design toward. Our ambition is to achieve a large increase in the translation rate of our primary screening experiments to downstream preclinical validation over the coming year.
Measuring endothelial cell age in the kidney
Our Vascular program is developing medicines to restore function in aged endothelial cells. Endothelial cells line the walls of our blood vessels, providing a transport system for nutrients and signals to flow into tissues. Endothelial cells also dictate which cells and signals can exit the circulation and enter a tissue at each juncture.
As we age, endothelial cells lose both their regenerative capacity and barrier function. The kidney is one of many tissues that suffers as a result. Kidney function declines in almost everyone with age, and age-related kidney diseases represent a massive burden on patients and the healthcare system. Kidney care accounts for roughly 0.5% of US GDP, higher than the GDP share of the Apollo Program. Our initial medicines are targeted at reprogramming renal endothelial cells to restore kidney function and address these needs.
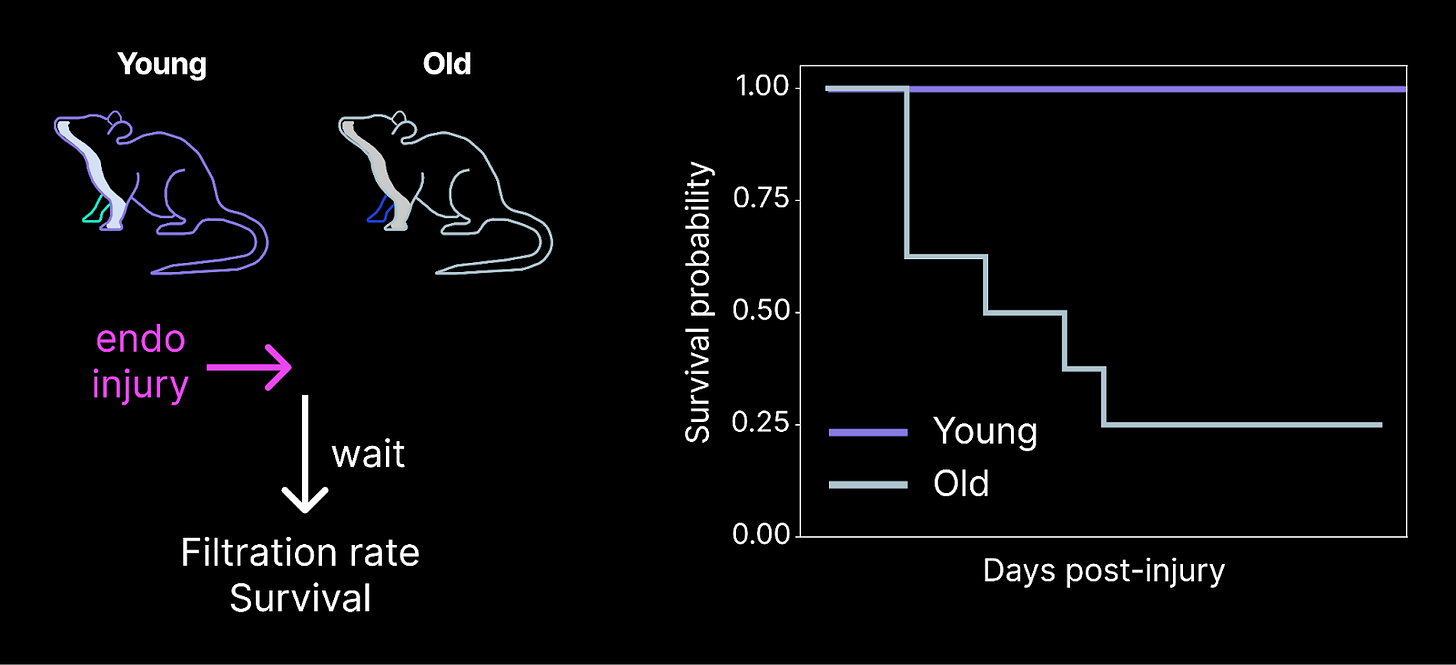
Before we can evaluate potential reprogramming medicines, we need to develop assays to measure age-related impairments in renal endothelial cells. The ideal assay demonstrates that old cells are less functional than young cells, failing to preserve health in the face of injury or disease.
Last month, we developed one such preclinical model that induces kidney disease specifically by injuring the renal endothelium. We discovered that old animals are significantly more susceptible to mortality from this injury than young mice. Given that these injuries are specific to the endothelium, these results suggest that old renal endothelial cells are significantly more vulnerable to injury than young cells.
Paired with our novel RNA delivery chemistry, we now have the tools required to begin testing reprogramming payloads for their effect on endothelial cell age.
Elsewhere
We’ve been privileged to share the NewLimit story in several venues recently. Find us elsewhere below!
Kleiner Perkins Builders podcast: we discuss epigenetic reprogramming, science-driven companies, and Chick-fil-A
Biotech TV: Brad Loncar interviews us inside the lab
Inc Magazine: NewLimit and our science are featured
Company growth
We continue to expand our team as we move toward the clinic. We’re recruiting for roles across the spectrum of seniority and across the research & development spectrum. Please reach out if you or a talented friend might be a good fit!


